- Blog
- About
- Contact
- Kids in the sandbox
- Battlefield 2142 single player weapon unlocks
- Protopie pricing
- Noteledge import pdf
- Linkassistant seo
- Walk behind string trimmer
- Aiseesoft free video converter mac
- Time digital clock
- Car town facebook game
- Itsycal reminders
- Definition of sonority in music
- Ublock origin for mac safari
- Canva slideshow
- Jelly jumpers nao baixa android
- Mla work cited sample page
- Pokemon emerald funny words
- Xml notepad xsd
- Corpse party seiko hentai
- Strife darksiders
- Whatsize for mac
- Hydrogen atom
- Pritunl vpn google
- Summa health surgery scheduler salary
- Linking iphone 6 to safari 5-1-10
- The postman book
- Ccleaner for mac


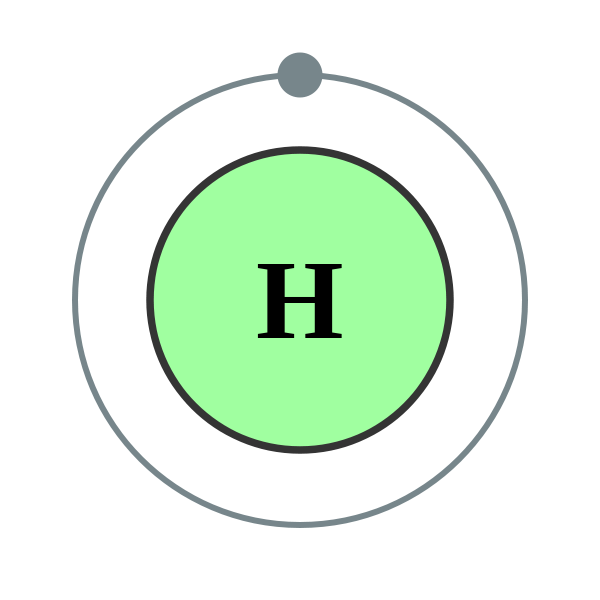
If you calculate the wavelength of this line using the energy expression from the Bohr theory, you get 656.11 nm for hydrogen, treating the nucleus as a fixed. The straight application of the Schrodinger equation to the hydrogen atom gives the same result. However, in most such cases the solution is not analytical and either computer calculations are necessary or simplifying assumptions must be made. As a class, models of the hydrogen atom are explored using an online java applet in this activity. The familiar red H-alpha line of hydrogen is a single line according to the Bohr theory. The Schrödinger equation also applies to more complicated atoms and molecules. The solution of the Schrödinger equation goes much further than the Bohr model however, because it also yields the shape of the electron's wave function ("orbital") for the various possible quantum-mechanical states, thus explaining the anisotropic character of atomic bonds. From this, the hydrogen energy levels and thus the frequencies of the hydrogen spectral lines can be calculated. The solution to the Schrödinger equation for hydrogen is analytical.
#HYDROGEN ATOM FULL#
Bohr's results for the frequencies and underlying energy values were confirmed by the full quantum-mechanical analysis which uses the Schrödinger equation, as was shown in 1925/26. Hydrogen has been around since atoms first formed in the residue of the Big Bang, and is the most abundant element by far. These assumptions, the cornerstones of the Bohr model, were not fully correct but did yield the correct energy answers. In 1914, Niels Bohr obtained the spectral frequencies of the hydrogen atom after making a number of simplifying assumptions. The first direct observation of the orbital structure of an excited hydrogen atom has been made by an international team of researchers. The energy levels are shown as numbers on the left side with the lowest energy level at the. The hydrogen atom has special significance in quantum mechanics and quantum field theory as a simple two-body problem physical system which has yielded many simple analytical solutions in closed-form. The figure below is an energy level diagram for a hydrogen atom. This article primarily concerns hydrogen-1. The most abundant isotope, hydrogen-1, protium, or light hydrogen, contains no neutrons other isotopes contain one or more neutrons. The electrically neutral atom contains a single positively-charged proton and a single negatively-charged electron bound to the nucleus by the Coulomb force. A hydrogen atom is an atom of the chemical element hydrogen.
